Energetics
Over the past 50 years our expertise has been providing new and innovative energetic materials solutions in applications where no other solutions previously existed.
We synthesize primary, high explosive and specialty chemicals, perform powder blending and pyrotechnic compounding and conduct propellant mixing, casting and machining. In addition, our R&D team develops new energetics for a variety of applications including high-temperature, product improvement and green drop-in replacements for existing primary explosives.
Our pyrotechnic materials transform energy to perform some kind of work when milliseconds matter, whether it be to actuate, detonate, generate, initiate, perforate, separate or terminate, you can rely on the necessary outcome you need to complete your mission. Energetic materials (EMs) have long been utilized for a variety of military, aerospace, commercial and oil and gas applications. While legacy materials deliver specific and established properties, there are circumstances and applications that demand new or customized EMs.
Whether you need to replace a legacy material that is no longer commercially available or have an application requiring new or modified EMs with enhanced properties— spanning reliability, sensitivity, stability, compatibility, output and safety—We can help. With a long history of EM synthesis and evaluation, chemists and chemical engineers on staff and dedicated testing and production facilities, we can help you with a range of EM needs.
Our energetics: A1A Ignition Mix, B/BaCr04 & WBKS, BKNO3, BNCP, DBX-1, Heat Powder Pyrotechnic Thermite, Hipel 710 Propellant Mixture, Lead Azide, THPP, ZPP, Lead Azide Replacements, KDNP.
Our chemistry facility is capable of receiving, analyzing, and performing safety testing along with fully documenting your projects which involve primary and secondary explosives
*NOTE: We do not sell our energetic materials.
About Our Energetics
A1A Ignition Mix

What it is: This pyrotechnic is an in-house favorite. It’s a blend of zirconium metal (fuel), red iron oxide (oxidizer) and diatomaceous earth (aka superfloss). We blend these by suspending them in solvent for safety purposes. The result is a fine dark red powder.
Why we like it: We’ve found that A1A works well when used as a transfer charge, transferring energy from a bridge composition to an output charge or another transfer charge.
There are several reasons why we like A1A as a transfer composition and why we think it deserves your attention:
- Generates very little gas
- Emits hot particles
- Thermally stable
- Long shelf life
We think this has an advantage over ZPP, another one of our pyrotechnic chemicals, because we use iron oxide instead of the oxidizer potassium perchlorate. Other benefits include low sensitivity to moisture and heat.
How to handle it: A1A is friction-, impact- and ESD-sensitive, which requires you to be smart about the way you handle it. First, understand that this energetic is one of the more electrostatic-sensitive chemicals in our facilities. Therefore, be vigilant and committed to the basics: only handle it with a velostat scoop and always confirm you are grounded. Second, you’ll want to wear typical personal protective equipment: smock, safety glasses and conductive shoes. If handling more A1A than normal, you may want to add additional safety measures to your PPE. Finally, do all your work behind a shielded and grounded work station. You don’t have to worry about detonation, but the rapid rate at which A1A burns results in a bright flash and audible crack that could cause burns or serious injury depending on how much chemical you’re working with. As for cleanup, make sure you blot the powder with a wet paper towel (50% IPA/DI). Do not rub the powder, as it is friction sensitive.
B/BaCr04 & WBKS


What they are: This powder duo is actually a pair of delay compositions: B/BaCrO4 and WBKS. B/BaCrO4 is made up of a boron(fuel) and barium chromate (oxidizer). WBKS uses tungsten as fuel and barium chromate and potassium perchlorate as oxidizers.
Why we like them: Like BKNO3, these pyrotechnic delay compositions have a wide variety of uses. B/BaCrO4 has a very rapid burn rate while WBKS is typically much slower. Giving us a wide variety of applications for their use. Their burn rate will vary by about 10% based on the ratio of components and the temperature at which they are used, which typically ranges from -65 to 160°
F. Additionally, WBKS is used in high temperature applications, particularly oil field exploration where its thermal stability allows use at 350° – 500° F.
How You Should Handle Them: In most cases, delay compositions are relatively stable. They possess low friction-, impact- and heat-hazard and have a moderate sensitivity to static hazards.
However, you’ll need to make sure you store these powders in extremely stable environments. Humidity and excessive jostling may cause inhomogeneity which can make the delays burn rate inconsistent. We advise storage of these materials in a desiccator.
The main hazard related to these chemicals is chromium, a toxic chemical that could cause cancer. As such, we recommend wearing your PPE and keeping any work area clean. Binder is added during manufacturing to minimize dusting when working with these compositions. There is a very low chance of accidental detonation or burning; the main concern is breathing in airborne particles
BKNO3

What it is: BKNO3 is a versatile energetic chemical with multiple uses at PacSci EMC. It’s a fuel-oxidizer mixture made up of approximately 24% boron, 71% potassium nitrate and 5% polyester binder.
BKNO3 may be purchased as either granular powder or prepressed pellets.
Why we like it: BKNO3 is an excellent pyrotechnic because of its stability—it’s not friction- or heat-sensitive and is only somewhat sensitive to static and impact. Other benefits of this energetic chemical include:
- Burn rate is minimally affected by ambient pressure
- Thermally stable
- Vacuum stable
- Generates gas and hot particles
- Long-lasting shelf life
As such, we use it with our gas generators and as an igniter for rocket motors. However, it can also be used in fuzes and certain propellants. BKNO3 is one of the only MIL-STD-1901A-compliant materials you can use for missile and rocket motor systems. Also, we like to use BKNO3 with laser initiated products.
How to handle it: Whereas A1A is less stable and can create more dangerous situations if something goes wrong, BKNO3 is less of a concern. In terms of storage, you’ll need to ensure you keep your BKNO3 dry. If it becomes too wet because of humid conditions or other situations, the moisture will oxidize the boron and degrade the chemical’s ability to emit energy. Wear your PPE when you’re working with this energetic. Though its toxicity levels are low to moderate, you want to make sure you avoid inhaling any dust in the area in which you’re working. In milligram and gram amounts, BKNO3 doesn’t pose any acute dangers. However, understand that, like A1A, this energetic won’t detonate but it burns rapidly and can produce sparks. When you’re cleaning up your work area, ensure that you flush it with 50% IPA/DI water and blot up the area with paper towels.
BNCP

What it is: BNCP is a relatively insensitive primary explosive made from sodium 5-nitrotetrazole, cobalt and potassium perchlorate. It was developed at PacSci EMC in conjunction with Sandia National Labs. It’s typically orange in color and both it and it’s combustion products are relatively non-toxic.
Why we like it: BNCP is a relatively stable (to 350 °F) material that is unique in that it is a DDT explosive. In order to function as a detonating explosive, it must be confined in plastic, aluminum or steel
housings/parts. In an unconfined state such as a loose powder, it will not sustain detonation. This makes it quite safe compared to other primary explosives during handling and loading. Once confined, it functions like any other primary explosive.
PacSci EMC uses BNCP in it’s FIREX line and it also finds applications in laser initiated ordnance where it is commonly doped with carbon black. PacSci EMC is the primary producer of BNCP and manufactures a variety of particle sizes for custom applications. Other manufacturers make it for research and development purposes, but don’t typically make BNCP in large quantities.
How to handle it: BNCP is a relatively insensitive energetic material and is not particularly static or heat-sensitive. It presents only mild friction and impact hazard compared to other primary explosives. Also, moisture, light and heat have minimal effects on BNCP. However, we suggest storing it in a closed puck and a low humidity environment. When you’re working with this material, use a velostat scoop and make sure you’re grounded. Proper safety wear includes a smock, safety glasses and conductive shoes. Cleanup of minor spills should be done with a 50% IPA/DI water mix and a wet paper towel for blotting. Like other EM’s, major spills require evacuation of the area.
DBX-1

What it is: DBX-1 is a primary explosive designed to function as a drop-in replacement for lead azide. It’s produced from copper(II) and sodium 5-nitrotetrazolate and it’s chemical name is copper(I) 5-nitrotetrazolate. DBX-1 was discovered and developed at PacSci EMC.
Why we like it: One of the best things about DBX-1 is that has no toxic or environmentally undesirable elements (like lead) that are used during manufacture, use or disposal. Because of this, DBX-1 is a great alternative to lead azide (LA) which presents a wide range of toxicity related health and safety issues. DBX-1 offers a “green” alternative to lead azide, and was designed to have physical and chemical characteristics, including output performance, sensitivity and thermal stability, that are nearly identical to those of lead azide. However, several DBX-1 characteristics that make it superior to LA:
- Has a better compatibility profile with other EM’s and ordnance components
- Does not decompose in non-hermetic systems
- Does not dead press at high loading pressures
- May be shipped wet under 2-propanol
How to handle it: DBX-1 is a very sensitive primary explosive and should be handled in the same way that lead azide would. It is very low friction, impact and ESD sensitive and is a point detonant.
Heat Powder Pyrotechnic Thermite
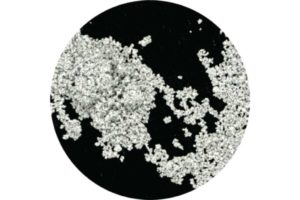
What it is: Heat Powder was developed as the pyrotechnic source in thermal batteries. It is a mixture of iron and potassium perchlorate (FE/KClO4) that is blended using a proprietary process to ensure consistent and reliable calorific output.
Our Heat Powder Pyrotechnic Thermite is designed with high burn rates for rapid activation of the battery and to provide low/no gas along with high pellet strength in order to maintain the mechanical integrity of the battery. As a replacement for Zirconium Barium Chromate, it improves storage life, mechanical properties, and ignition speed. it can be tailored to meet the needs of your program.
Why we like it:
- No/little Gas Generation
- Dimensionally Stable After Ignition
- Easy Ignition
- Excellent Long Term Stability
- Consistent Calorific Output
- High Burn Rate
- Good Pellet Strength
How it works: Used in thermal batteries as both the electrolyte and the heat source, because of the stability of the material, these batteries have a long shelf life with no degradation. Once the powder is activated the battery provides powder for the required application. Our heat powder is designed with high burn rates for rapid activation of the battery and to provide low/no gas along with high pellet strength in order to maintain the mechanical integrity of the battery.
Hipel 710 Propellant Mixture

What it is: Hipel 710 Propellent is used in the oilfield, when mixed in a 50/50 blend with Hipel 3500 will deflagrate and propel the coring bullets at their optimum velocity is obtained. Hipel 710 Propellent, a trade name for a hydroxyl-terminated polybutidiene (HTPB) propellant, comprising 84% ammonium perchlorate (NH4 ClO4) (oxidizer), 9.1% HTPB (fuel), 0.7% aluminum trihydrate (Al2 O3 x3H2 O) (burn rate catalyst), 0.2% Fe2 O3 (burn rate stabilizer), and 6% polymide (Kermid 601).
Why we like it:
- Operating Temperature: 360°F for 12 hours
- Output: Deflagration output of 1215 +- 40 calories / GM
- Shelf Life: 10 Years when stored unopened in original packaging
How it works: This is a readily ignitable propellant mixture. The mixture is sensitive to ignition by flame, heat, friction, impact or electrostatic discharge. When ignited, the mixture burns producing heat, flame, hot gasses and hot particles. It offers a long shelf life and high stability. Since it is a simple composite propellant, it has less volatile contaminants than other available gun propellants.
Hipel 710 has a long history of use by a leading provider of oil & gas services.
Lead Azide

What it is: Lead azide (LA) is a white primary explosive composed of lead and nitrogen. Three common varieties of LA are produced based on different additives: dextrin (DLA), polyvinyl alcohol (PVA) and CMC (RD-1333). Manufacture with these additives optimizes the crystal shape so that the LA will flow through automatic loading equipment. The additives also serve to reduce the sensitivity of the lead azide.
Why we like it: LA is a very versatile energetic material. We typically purchase inexpensive LA from an outside source but will manufacture high sensitivity LA for unusual applications. At PacSci EMC it is used as an initiating agent, as a transfer charge and in stab primer applications. Some of the explosives we use it with include:
- HNS
- RDX
- CH-6
- PETN
What makes it such an effective primary explosive is that it detonates quickly, is thermally stable and has a long shelf life in hermetic applications.
How to handle it: LA is an incredibly sensitive explosive. It is one of the most sensitive materials we use at PacSci EMC. To give you an idea of exactly how hazardous it can be, its sensitivity levels for friction, impact and ESD are:
- Friction: ≤ 10 grams
- Impact: ≤ 30 centimeters
- Static: ≤ 7 megajoules
Despite having enhanced sensitivity, LA is widely used at PacSci EMC and many other ordnance manufacturers every day without issue. You’ll want to use the standard PPE procedure when working with this material: smock, safety goggles and conductive shoes. All work should take place behind a shielded workstation. You should use additional PPE when you’re dealing with a large quantity of LA. Lead azide isn’t sensitive to light, heat or moisture. Therefore, storage rules are straightforward: Keep in a puck in low-humidity environments.
THPP

What it is: THPP is a “booster” pyrotechnic and is a blend of a fuel (titanium hydride. TiH2), an oxidizer (potassium perchlorate, KClO4) and binder, typically Viton. The primary use at Pac Sci EMC is as a pyrotechnic transfer composition where it converts the heat energy from a bridgewire pyrotechnic (usually ZPP) to generate a flame and hot particles which ignite an output charge. THPP affords a rapid pressure rise, emits hot particles and has good thermal stability.
Why we like it: THPP was designed to be much safer (from an ESD perspective) to use during manufacturing operations compared to TiKP which contains titanium metal (similar to ZPP) and is extremely ESD sensitive. THPP also has a lower ignition sensitivity and slower burn rate compared to TiKP and, while both materials are used in ordnance devices, they frequently have different applications. THPP is prepared at Pac Sci EMC from commercially available titanium hydride powder, finely ground potassium perchlorate and a small percentage of Viton binder utilizing a shock gel mixing method.
How to handle it: Like ZPP, THPP requires both assembly personnel and hardware used in manufacturing processes to be efficiently grounded and have extensive PPE due to the ESD sensitivity of the composition. THPP, both bulk and pressed, has been found to be insensitive to initiation from a “typical” human body static discharge and so is considered safe relative to TiKP or TiH<2PP (titanium or titanium subhydride). Minor compatibility issues between THPP and typical EED materials have been found. These includes various amines commonly found in curatives for adhesive resins and common solvents.
Of note is that THPP is not as thermally stable as other common pyrotechnics encountered at Pac Sci. Long term exposure at high temperatures (>250 °C) may alter the titanium/hydride ratio which changes the safety and output properties as well as the service life of components containing the composition.
Cleanup of minor spills should be done with a 50% IPA/DI water mix and a wet paper towel for blotting. Like other EM’s, major spills require evacuation of the area.
ZPP

What it is: The pyrotechnic zirconium/ potassium perchlorate (“ZPP”) is a mixture of fuel (high purity micron range Zr powder) and an oxidizer (milled potassium perchlorate). ZPP has been widely used in electro-explosive devices for many years as a composition that converts the electrical energy applied to a bridgewire into sufficient heat and hot particles to initiate a downstream transfer charge or explosive output. ZPP is used in defense, automotive initiators and in the NASA Standard Initiator where it plays a critical role in initiating various pyrotechnic events in space applications. ZPP has the advantages of affording a rapid pressure rise, generating only minor gas, emitting hot particles, being very thermally stable (>450 °C) and having a long shelf life.
Why we like it: Due to the demanding nature of applications that utilize ZPP it has been extensively investigated and the chemical, physical and output properties of mixtures with a variety of stoichiometries are well known. Under the right conditions ZPP can meet the 1 amp/ 1 watt/ 5 minute no-fire safety requirement imposed by U.S. military design specifications (e.g., MIL-I-23659). This is partially due to the high thermal conductivity of ZPP mixtures that effectively move heat away from the bridgewire and into the bulk material under no-fire conditions. In addition, ZPP has been demonstrated to function normally at both extremely high and extreme low (cryogenic) temperatures when manufactured and loaded under optimum conditions. In more extreme environments ZPP may be utilized as a bridgewire slurry to insure consistent ignition. ZPP is electrically conductive due to the zirconium content, which renders it particularly vulnerable to electrostatic discharge and which makes it a safety hazard during handling and loading in ordnance items. ZPP is typically produced with ~5% Viton B as a binder and may contain smaller percentages of graphite as well. Viton is incorporated to provide some level of electrostatic sensitivity protection while the purpose of the graphite is to act as a lubricant to increase packing density and as a reaction rate modifier. At Pac Sci EMC, ZPP is typically prepared via a wet process which affords safety benefits over other methods of production while producing a product that is extremely homogeneous and consistent.
How to handle it: ZPP is not particularly sensitive to moisture but should be stored in a low humidity environment. Components containing ZPP are typically hermetically sealed and can have a long operational lifetime. The extreme ESD sensitivity of ZPP requires both assembly personnel and hardware used in manufacturing processes to be efficiently grounded and have extensive PPE. Even with
safeguards in place, inadvertent ignition of ZPP mixtures are not uncommon and may produce an audible crack along with an intensely bright flash.
APD personnel at PacSci EMC are currently engaged in development of an ESD safe non-conductive replacement for ZPP called BI-820. If successful, this mixture of organic fuel and
oxidizer, in optimized ratio, may provide an output similar to other pyrotechnic systems, but would have the benefit of being non-conductive both prior to and after ignition as well as being
stable at very high temperatures. Details related to BI-820 may be found in U.S. Patent 11,878,951.
About our Green Energetics
When it comes to energetic materials, you know that an environmental impact is inevitable. Our Green Energetics take impact into consideration.
After all, it’s the nature of the work you do with detonators, primers, ignition devices and pyro train elements each day. And although “greener,” lead-free energetic solutions with less impactful disposal and detonation methods have recently emerged on the market, they aren’t always as effective at delivering the results you require to produce at high volume and stay competitive.
We’ve spent two decades engineering alternatives to existing lead azide and lead styphnate materials that don’t just meet industry standards for cost-effectiveness and efficiency—they exceed them, in both defense and commercial applications.
Lead Azide Replacements
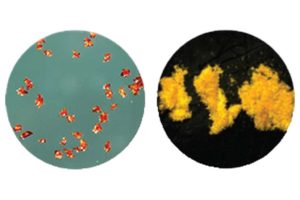
DBX-1 is a copper-based drop-in green energetics replacement for all variants of lead azide (LA) with potential to replace DXN-1. DBX-1 has energetic characteristics that our industry has been trying to duplicate for the last 50 years.
All variants of lead azide (LA), including RD-1333, Dextrinated Lead Azide, and PVA have been widely used in ordnance systems for many years. Nearly all chemical detonators use LA as the initial shock wave-generating compound for proper detonation of subsequent main explosive charges.
We completed an 8020.5C military qualification program in 2009. DBX-1 was successfully evaluated against LA in many products across the spectrum of defense and commercial applications. Other companies (including several DoD organizations) utilizing LA evaluated DBX-1 in a variety of applications with very positive results.
We synthesize DBX-1 in 100-gram batches for use by government and industry. Future scale-up to 1 Kg and beyond is underway with support from the US Army and commercial organizations.
Lead Styphnate Replacements
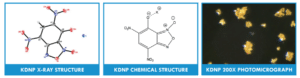
Lead styphnate (LS) is a primary explosive used in ordnance systems as components in defense initiation trains. These applications include non-corrosive percussion primers, stab initiated devices, bridgewire initiated compositions, and detonators, and is used in nearly all commercial primer applications as well. Lead styphnate has been widely used in ordnance systems for many years.
PacSci EMC completed an NAVSEAINST 8020.5C evaluation testing on an LS green energetics replacement, KDNP (4,6-dinitro-7-hydroxy benzofuroxan, potassium salt. Based on the results, Naval Sea Systems Command qualified KDNP as a primary explosive in accordance with the requirements of NAVSEAINST 8020.5C and identified the compound safe and suitable for service use, and qualified for weapons development.
In addition to qualification testing, KDNP (U.S. Patent 8,062,443 for use in Lead Free Primers) successfully evaluated LS performance in a variety of applications. KDNP has compatibility equal to – or greater than – LS with a variety of ordnance materials and has substantially greater output on a weight basis in pressure-time (closed bomb) testing. KDNP may be recrystallized to a particle-size, appropriate for use in a variety of systems to be tailored for use as either a small particle component of a bridgewire slurry or a large particle constituent of a stab primer.