DESENSITIZED RDX DUE TO CRYSTAL GROWTH
RDX loading density of greater than 98% TMD (3) created conditions for recrystallization of RDX (from released occluded cyclohexanone) into large crystal structures. These large RDX crystal structures are void less, therefore not allowing Hot Spots for a shock stimulus to generate the conditions needed to start and sustain a chemical reaction.
Robert Weinheimer
Published: 27th International Pyrotechnics Seminar
Table of Contents
Abstract
GIDEP ALERTs in 1976(1) and again in 1988(2) reported RDX detonating cord and linear shaped charge (LSC) failures to initiate, and mid-propagation failure after initiation. This paper is a report on time dependent desensitization of RDX due to crystal growth induced by batch process residual solvents, acetone, and cyclohexanone.
Linear Shaped Charge (LSC) failures were experienced during customer incoming inspection acceptance testing six weeks after successful LAT at the manufacturer. The RDX desensitization was attributed to RDX recrystallization some time after high density loading during assembly and after LAT at the manufacturer. To understand the cause of failure, RDX literature review, and test analysis are discussed. The 1976 GIDEP ALERT identifies the recrystalline solvent to be occluded acetone while the 1988 GIDEP ALERT identifies occluded cyclohexanone was the recrystalline solvent. This paper is based upon the 1988 investigation results.
CONCLUSION: RDX loading density of greater than 98% TMD(3) created conditions for recrystallization of RDX (from released occluded cyclohexanone) into large crystal structures. These large RDX crystal structures are voidless, therefore not allowing hot spots for a shock stimulus to generate the conditions needed to start a chemical reaction.
THEORY: 1. High density RDX will fail to initiate when the length of the hot spot (void) is less then the length of the shockwave pulse. ie:
A – If the shockwave pulse in the RDX is overcome by the rarefaction wave in 4mm, then a void greater than the length of the shockwave, must be within the range of the shockwave pulse (<4mm) for the RDX to initiate
B – The RDX will not initiate if the shockwave pulse is attenuated to a pressure wave before reaching a hot spot void.
Failure Modes
10 GR/FT DETONATING CORD
In 1982 the Navel Weapons Support Center reported RDX crystal growth during a Scanning Electron Microscope examination of 10gr./ft RDX (with Duponol G & Rhodamine B) cores of lead sheath Detonating Cord. A prime defense contractor conducted an investigation on samples of the same material and duplicated the NWSC examination with similar findings.
15 GR/FT LINEAR SHAPED CHARGE (LSC)
In 1985 Lockheed Missiles Systems Company reported RDX crystal growth in manufactured 15 gr/ft RDX LSC (aluminum sheath). The contractor’s analysis of crystal growth due to residual solvents was substantiated by SNL analysis.
FAILURE MODE
Many explosives are known to have the characteristics known as “Dead Pressing”. RDX is one of them. Dead Pressing (as used in this report) is when an explosive is consolidated to a very high density greater than 98% TMD (1.763 gm/cc for RDX). RDX has a direct TMD of 1.799 gm/cc and an X-Ray TMD of 1.806 gm/cc(3). Densities greater than 98% Direct TMD (1.76 g/cc) reduce the interstitial voids and desensitizes RDX to initiation by shock stimulus.
LSC ALUMINUM SHEATH
The tubing is bulk loaded with RDX and then drawn through funnel dies to reduce the diameter. This reduction in diameter increases the density of the RDX explosive. The tubing is then swaged through a series of hammer swaging dies which again reduces the tubing diameter and finally a hammer die upsets the tube into a chevron shape (inverted “V”) with a desired average density of 1.75 g/cc.
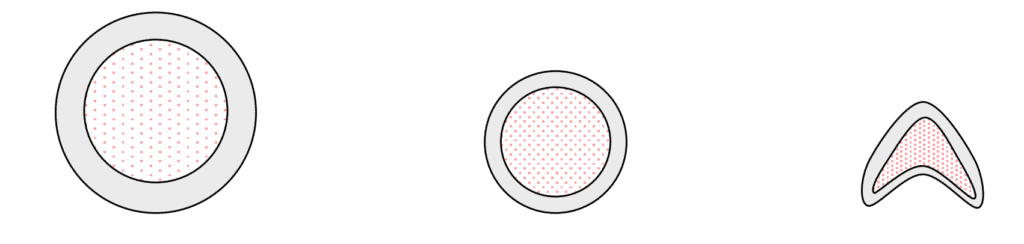
Under high density, the RDX crystals are forced closer together. The RDX particles are crushed releasing trapped (occluded) cyclohexanone and fill, or reduce the volume of the interstices (voids). The released cyclohexanone recrystallizes the crushed RDX into larger crystal structures. When the crystal structure reaches a size greater than the shock wave duration to the nearest void the propagation stops or never gets started. Thus the reduced voids in the loaded explosive desensitized the RDX.
The RDX used in the LSC was treated with Duponal G as a binder and Rhodamine B as a coloring agent. The coloring agent was used to differentiate from other explosives (HMX, HNS, PETN) used in the manufacture of LSC. Rhodamine B is a red coloring agent in concentration but when diluted with the RDX it renders the explosive powder Pink in color. The two additives were also added as benign contaminants that would help discourage recrystallation after LSC manufacture. Observation of the RDX at the area of the LSC which failed to propagate had a red periphery and not pink as expected.
This leads to the following theory on the increased red color from the original pink powder:
As the RDX crystal grows it displaces the additives (Duponal G and Rhodamine B) forcing the viscous material to areas where there are voids. This causes the additives to migrate to the outside of the crystals in higher concentration. Thus, the appearance of increased redness at the periphery of the RDX in the area of failure.

The RDX crystal fractures at densities above 98% TMD releasing occluded cyclohexanone. The fractured crystal growth begins at the point of highest density and radiates away from the density center. In the case of LSC the highest density would be expected at the apex of the chevron upset. The pink Rhodamine B previously evenly distributed throughout the RDX is displaced to less dense areas of the RDX. The increased concentration of Rhodamine B becomes a deeper red observed at the periphery of the recrystallized RDX.
During the recrystallization process crude RDX is recrystallized using cyclohexanone which has a boiling point of 156°C (acetone has a BP of 51°C by comparison). The high boiling point of cyclohexanone makes it difficult to remove from the RDX. HMX uses acetone as the recrystallization solvent that can easily be driven off.
Hot Spots
There are three interacting theories discussed in the literature review on the impact or mechanical initiation process, Hot Spots by adiabatic compression, friction and the more accepted effect, inelastic flow.
Adiabatic Compression
The spaces (interstices) between the RDX crystals are voids filled with air (gas). An initiating shock wave upon entering a void creates adiabatic compression of the air thereby generating very high pressure and temperature (Hot Spot). The temperature exceeds the autoignition temperature of the explosive causing a chemical reaction. This theory, if it were correct, would predict failure of an explosive to initiate in a vacuum. A shock wave can not propagate through a vacuum. Therefore, adiabatic compression of Hot Spots could not be achieved in voids between crystals. Yet, RDX, HMX, and HNS are known to be capable of initiation and propagation in a vacuum.
Friction
1) The interstices, as voids between RDX crystals, offer no resistance to crystal fissure slip planes. When a shock wave dislodges a crystal along its fissure the friction along the slip plane creates the hot spot temperature needed to autoignite the RDX.
2) The interstices, as voids between RDX crystals, offer no resistance when a shock wave dislocates an element from the RDX molecule. The imbalance of the molecule oscillates a chain reaction to surrounding RDX molecules generating the temperature required to start and sustain a reaction for initiation and propagation.
Dislocation theory allows initiation and propagation in a vacuum.
The adiabatic compression and friction theories may be interdependent in establishing the sensitivity of the initiation threshold level of an explosive. Some explosives have been noted to be less reactive in a vacuum. The dislocation theorists say that when the air is present it enters into the chemical reaction making the detonation more violent.
Inelastic Compression
The inelastic compression theory is the most accepted mechanism for initiation and sustained propagation of explosives. Under shock or mechanical impact the solid explosive particle would be under extreme compressive, shear, and tensile stresses. Under high-pressures the explosive melt point increases therefore not limited by the melt phase stability seen at 1 atm(4).
Particle Size: Intragranular particle size can affect the sensitivity of an explosive considerably. PETN, RDX, HMX, HNS, & RDX are known to become increasingly sensitive as the particle size decreases. However, there is a reversal transition point in the lower particle size range where sensitivity begins to decrease again. It is not the particle size by itself that influences the sensitivity but the particle size relative to the size of the voids. The critical diameter of a “hot spot’ is between 10-5 and 10-3 cm(4). Shock sensitivity will decrease with the decrease of void size as the void size approaches the width of the shock front (typical 4µm for HNS @ 30 kbar)(5).
Void Collapse: Thermal energy release in a Hot Spot is dependent upon void volume. A shock front dislodges RDX particles as it enters the void and carries the particles across the void to impact the downstream RDX surface. The thermal energy is generated by the friction from particle dislodging, void air volume compression, and the surface impact mechanism. An increase of thermal energy release is experienced as the void cross section approaches that of the shock front width(5).
Sieve analysis of RDX at LMSC Santa Cruz has determined that percentage (by weight) of occluded cyclohexanone increased with particle size. The percentage of HMX increased with the decrease of particle size.
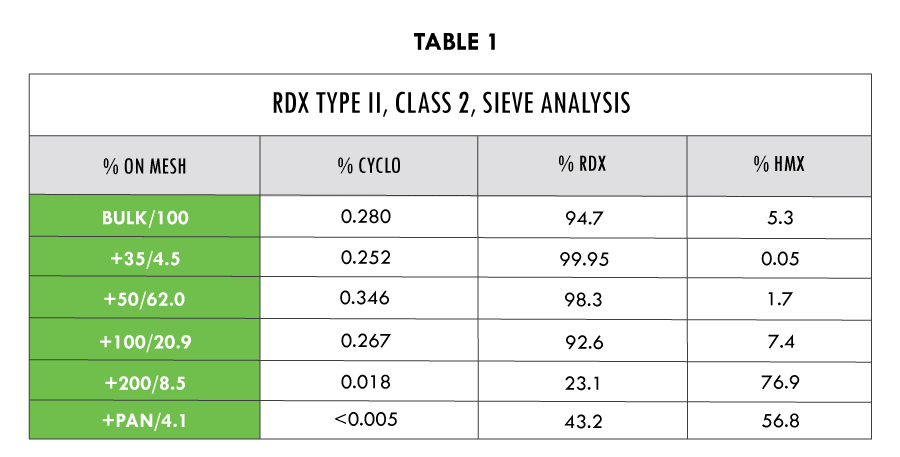
The table data is taken from two different sample analyses and combined to represent a general expectation for a particular sieve mesh size. The percent cyclohexanone was taken from a different sample analysis then the RDX/HMX percent ratio. However, both sample sets of data are combined by a common mesh size.
Discussion
Hexamine is one of the starting materials used in the production of the explosives RDX and HMX. Hexamine is a reactive heterocyclic fused ring compound produced by condensation of formaldehyde and ammonia.
RDX and HMX are manufactured at the Holston Army Ammunition Plant (HAAP). The plant is located in Kingsport, TN on the Holston River.
Both RDX and HMX are used in a large number of explosive mixtures which have a variety of end uses. RDX and desensitizing waxes are designated as Composition A explosives. These mixtures are used in press loaded small artillery shells. Compositions A-3 and A-5 are also used as booster charges in place of Tetryl which is no longer manufactured by the Army.
Composition B and Cyclotols are mixtures of RDX and TNT. Desensitizing wax is also added to the Composition B products. The superior properties have lead to the extensive use of Composition B as] burster charges in bombs, artillery shells and land mines(6). For this purpose, the Composition B is melt loaded into the end item. Once solidified, the cast explosive can be drilled for insertion of non-bursting charge components.
Other major groups of explosive mixtures containing RDX are the plastic bonded explosives(7) and demolition charges. These formulations are known as Composition C, PBX, and PBXN explosives and contain RDX compounded with plasticizers and solvents.
Cyclohexanone is used as a recrystallization solvent in the manufacture of RDX at Holston. At the 1978 production level, Holston utilized 180,000 lb. of cyclohexanone. The civilian cyclohexanone production is 1.6 billion lb. per year. The major civilian use of cyclohexanone is as a chemical intermediate in the manufacture of nylon 66 and nylon 6. It is also used as a solvent and in the manufacture of caprolactone. In the environment, cyclohexanone is degraded by photochemical and microbial pathways.
Holston has ten RDX/HMX lines capable of producing 250 million lb./yr. of RDX and 15 million lb./yr. of HMX at full capacity. In 1978 Holston was operating at only 8% capacity.
References
Appendix I: Linear Shaped Charge Photographs
Appendix II: Energetic Materials Supplementary Tables
RDX and HMX DENSITY VERSUS VELOCITY OF DETONATION